“It’s 8.44 milligrams like last time, and you’re to have the dose over one hour,” she says. She looks towards the digital clock on the wall.
“The time is now 12:08 p.m., so—in exactly 15 minutes—I’ll be back to measure your heart rate and blood pressure.”
The patient nods in agreement and the nurse’s right finger settles calmly on the start button of the medicine pump.
“All right, here we go,” she says and pushes the button.
The patient in room 29 suffers from incurable malignant pleural mesothelioma as a result of a long life with insolation work. Asbestos fibres have deposited themselves in his lung tissue, and neither chemotherapy nor surgical surgery has been able to remove the cancer. Together with 30-50 other terminally ill cancer patients, he therefore participates in a medical trial of the DTU-patented drug MBS8 from the company MonTa Biosciences.
Tests on animals have shown that the drug can activate the animals’ immune system, so that the immune cells themselves begin to fight the cancer. Now the patient trials at Rigshospitalet and Herlev Hospital are to show whether the same applies to humans.
“In the phase 1 study that is running now, we need to find out whether the drug is safe and establish the right dose. When this has been done, we can proceed to phase 2 of the study, where the focus is on efficacy,” explains Kristoffer Rohrberg, Chief Consultant and Head of the Phase 1 Unit at Rigshospitalet’s Department of Oncology.
On Wednesday, 21 April 2021, he treated his first patient with the drug from MonTa Biosciences. On the same day, MonTa CEO Simon Skjøde Jensen called the event a milestone.
A weapon against side effects
It was not a milestone that Simon Skjøde Jensen had expected to reach when— more than ten years earlier—he worked as group manager in the pharmaceutical company Bioneer in Hørsholm.
“If I had known then, where I would be today, I don’t think I would have believed it,” he says, seeing that only 40-50 medical studies a year end in trials at Rigshospitalet. And only one or two of them are Danish.
"If I had known then, where I would be today, I don’t think I would have believed it"
Simon Skjøde Jensen, CEO and co-founder of MonTa Biosciences
In fact, Simon Skjøde Jensen’s focus was not on patient trials when he started up a research project back in 2007 with Thomas Andresen, current Professor at DTU Health Tech. Nevertheless, the meeting with Thomas Andresen was decisive for the subsequent formation of MonTa Biosciences. The two researchers realized that—with their combined knowledge—they could solve a known problem in immunotherapy: that treatment with immunotherapy often causes serious side effects.
The reason is that a direct injection of an immunostimulant in the blood triggers an extensive activation of the body’s cells. The drug activates both the desired neutrophils that are the immune system’s effective disease fighters, while also activating the other cells and blood vessels of the body, which may result in serious side effects. This means that the dose connected with immunotherapy must often be kept to a minimum, which reduces its efficacy.
Developed lipid ball
In the DTU laboratories, Simon Skjøde Jensen, Thomas Andresen, and a team of researchers and PhD students therefore began developing a special lipid ball technology aimed at solving the problem of the serious side effects. The lipid ball they developed is a nanoparticle that carries the immunostimulant safely to its target without impacting the rest of the body. When the lipid ball is injected into the blood, it thus transports the immunostimulant directly to the neutrophils without causing undesirable activations. The neutrophils absorb the substance and their internal signal receivers—the receptors—are activated.
“The cells are informed that there is a disease in the tissue, and they are programmed to enter the tissue and fight the disease. For cancer patients with solid tumours, it’s precisely the tissue that has been affected, so—with this technology—the neutrophils enter the tissue and start breaking down the tumour,” explains Simon Skjøde Jensen about the technology for which he holds a patent today.
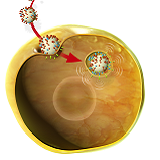
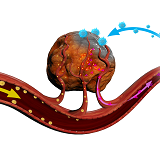
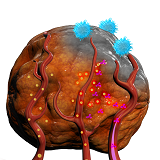
How MBS8 activates the body's immune system:
 |
 |
 |
 |
The drug from MonTa Biosciences, MBS8, is injected into the bloodstream and is transported directly to the body’s neutrophils, which are among the disease-fighting cells of the immune system.
|
The neutrophil absorbs the active substance—the TLR7 agonist—of the drug.
The substance binds to the cell’s internal TLR7 receptors— which act as signal receivers—and the cell is activated. |
The activated neutrophils are informed that there is disease in the tissue. They consequently migrate to the site of the disease via the blood vessels of the body. By ‘rolling’ along the vascular wall, the cells find a point of access in the vascular wall. From here, they go through the vascular wall and enter the tissue of the tumour.
The neutrophils excrete toxic substances which kill the tumour cells. This results in the tumour releasing tumour antigens, which are proteins produced by the cancer cells. |
The release of antigens in the tumour causes an immune memory response in the body’s other disease-fighting cells, namely the blue memory T cells found in the lymph nodes. This means that the T cells can now also recognize the tumour cells that they are helping to kill.
The effect starts about a week after the treatment with MBS8 and continues long after. This contributes to curing patients with metastasized cancer. |
In 2014, the invention resulted in the formation of the biotech company MonTa Biosciences, and three years later the last piece fell into place. Here, MonTa’s partner—a pharmaceutical company in Switzerland—terminated their licence agreement for a promising immunostimulating drug that could be used for the researchers’ lipid ball: a molecule called a TLR7 agonist. The researchers applied for the license—and got it.
Finally, they were able to start studies on animals and find the investment needed to reach the hospitals’ trials sections.
Email from Rigshospitalet
Every Monday and Wednesday afternoon, Simon Skjøde Jensen and his colleagues are ready in front of the screens in DTU Science Park, where their office is located. In fact, Monday and Wednesday are treatment days, and this means an email from Rigshospitalet’s Department of Oncology.
The email contains data showing how the MBS8 treatment has gone in the patients, and—in DTU Science Park—the data are eagerly compared with the results from the previous animal trials.
Trials with MBS8 in mice have shown—among other findings—that the neutrophils of the mice begin to enter the tissue within six hours after the injection and that they have killed up to 80 per cent of the tumour cells in the course of 24 hours. It cannot yet be ascertained whether the effect on the patients’ tumours is the same, but the reaction in the patients’ immune system follows that of the mice.
"So far, it looks like there is the same reaction in the patients as in mice. In blood samples from the trial patients, you can see that the way in which the immune cells are activated is exactly the same as in mice,” says Simon Skjøde Jensen. He is also pleased that the side effects in the patients have been mild—as expected.
The sender of the weekly emails—Chief Consultant Kristoffer Rohrberg—is equally impressed and puzzled by MonTa’s performance. He has 16 years of experience with phase 1 studies at Rigshospitalet, but he has not previously come across a similar company.
“I have a special feeling about this. Their work has been so convincing, especially considering that they are a completely new company. Even during the coronavirus crisis, there were no hitches. The time frame was only delayed when we were to start testing the drug here, which is really unusual,” says Kristoffer Rohrberg, who had to request a postponement of the start of the patient trials with MBS8.
Kristoffer Rohrberg is used to the companies themselves postponing the time of commencement. He had therefore not expected MonTa Biosciences to be ready on time. But they were.
New hope
The time is observed at Rigshospitalet’s room 29. The digital clock on the wall says 12.23 p.m., and nurse Christina Sass-Petersen enters the room.
“The fifteen minutes are up,” she tells the patient, grabbing the cuff on the sphygmomanometer on a table next to his bed. She opens the flap and secures the cuff to the patient’s left arm.
“I will just check up on you,” she says, and starts measuring blood pressure and heart rate.
At the foot of the bed sits the patient’s wife, who follows the procedure attentively. She is there every Monday when he is treated, she listens every Friday when he talks on the phone with the consultant, and she is there when he has blood tests and scans done every fourth week. Both she and her husband were given new hope when they were presented with the trial with the drug from MonTa.
“It gave us a feeling that they haven’t given completely up on me,” her husband explains.
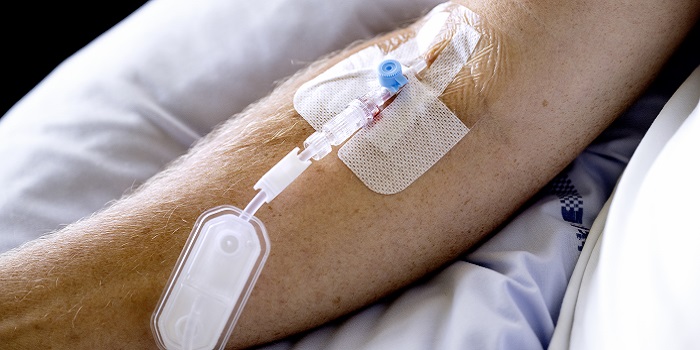
The phase 1 study with the cancer medicine MBS8 has given terminally ill cancer patients new hope. (Photo: Bax Lindhardt)
The effect on his cancer disease and on the other phase 1 patients’ cancer is not expected to be ascertainable until the start of 2022, and there will then be some way to go before the drug can be finally approved for treatment.
After the phase 1 studies—which will run for another couple of months—efficacy on cancer tumours must be documentable in phase 2, and, finally, a phase 3 study must document a survival gain in the patients.
The journey has been long, and it still is. Nevertheless, Simon Skjøde Jensen has no doubt about what drives him.
“I have friends who have died of cancer, and every time I recall them, I think: What I’m devoting my life to makes sense. By now, we’ve put many hours into this, and when the going has been tough, I've always returned to our basic mission: We want to make the world a better place for those who become terminally ill with cancer.”
For the sake of the scientific trials and for ethical considerations of relatives, the patient appears anonymously in the article.
Read more: www.montabiosciences.com